https://doi.org/10.1021/acs.inorgchem.5c05869
(Accepted February 2026, Published February 2026)
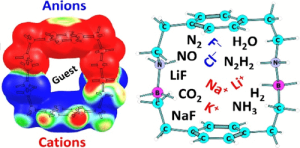
Azaboracyclophanes constitute a new class of three-dimensional, charge-separated molecular cavities in which B–N dative bonds function as internal polarizing elements, generating oriented electrostatic environments akin to confined electric fields. Using density-functional theory, we show that systematic incorporation of B–N linkages into cyclophane scaffolds produces large and programmable intramolecular polarity, shortens B–N distances, and amplifies internal MESP minima by up to ∼ 3× relative to carbon analogues. This electrostatic engineering profoundly reshapes host–guest behavior: azaboracyclophanes strongly stabilize cations and anions on both outer surfaces and within the cavity, convert a weakly binding parent framework into an ambivalent receptor, and enhance internal binding of polar guests by up to 57%. Distinct BN-placement motifs (NN7BB vs NB7BN) enable selectivity switching, including preferential stabilization of quadrupolar CO2. These results establish B–N-doped cyclophanes as programmable supramolecular “reaction chambers,” where electric-field-like effects emerge from molecular design rather than external application. The principles revealed here suggest general strategies for ion recognition, gas capture, and field-assisted reactivity within polarizable nanocavities.