https://doi.org/10.1039/D6OB00128A
(Accepted February 2026, Published February 2026)
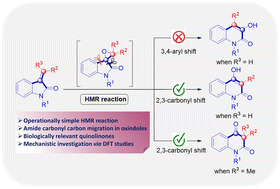
House–Meinwald rearrangement of oxindole substrates facilitating a one-pot synthesis of 3-substituted 4-hydroxyquinolin-2-ones and quinoline-2,4-diones under EBHP and base-mediated conditions is reported. This transformation proceeds through a well-defined sequence involving epoxidation, formation of a quinone methide-type intermediate, and subsequent ring expansion. The crucial carbonyl migration step was further substantiated by DFT studies. Moreover, the synthetic utility of the resulting 3-substituted 4-hydroxyquinolin-2-ones was demonstrated through late-stage diversification, highlighting the versatility of this protocol.