https://doi.org/10.1021/acs.jpca.5c08587
(Accepted February 2026, Published February 2026)
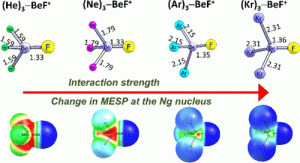
The interactions of noble gases (Ng = He, Ne, Ar, Kr) with highly electron-deficient main-group fragments are systematically investigated by using high-level CCSD and CCSD(T) calculations. A broad set of electrophilic acceptors is considered, spanning six-electron (O, S, F+, Cl+, Br+, OH+, SH+, NH2+), four-electron (BF2+, AlF2+), and two-electron (BeF+, MgF+) centers. Optimized geometries, interaction energies, and electronic descriptors reveal a continuous evolution of Ng binding behavior across the series from weak polarization-dominated interactions for He and Ne, to donor–acceptor bonding for Ar, and to strongly covalent-like coordination in Kr complexes. The analysis, supported by natural bond orbital (NBO), quantum theory of atoms in molecules (QTAIM), symmetry-adapted perturbation theory (SAPT), and molecular electrostatic potential (MESP) descriptors, demonstrates that the strength and multiplicity of Ng binding are governed primarily by the electrophilicity and valence-electron deficiency of the acceptor fragment with noble-gas polarizability modulating the interaction strength. Within this context, a unified 2e–4e–6e valence-electron framework is employed as a descriptive tool to rationalize why six-electron centers preferentially bind one Ng atom, four-electron centers stabilize two Ng atoms, and highly electron-deficient two-electron centers accommodate three Ng atoms. Among the multi-noble-gas complexes examined, BeF+ and MgF+ are found to stabilize tri-noble-gas adducts across the He–Kr series, with Kr3BeF+ exhibiting the strongest overall binding. Trihelium coordination to BeF+, with interaction energies of several kcal mol–1 per He atom, highlights the remarkable stabilization that can arise in extreme electron-deficient environments. Overall, the results provide a unified and internally consistent framework for organizing mono-, di-, and tri-noble-gas binding motifs across the noble-gas series, clarifying the electronic factors that govern noble-gas coordination in highly electrophilic chemical regimes.